Understanding Hybridoma Technology in Monoclonal Antibody Development
Hybridoma technology is a proven and powerful method that combines an antibody-producing B-cell with a myeloma cell, creating a hybrid cell line known as a hybridoma. These hybridomas can produce large quantities of identical, or monoclonal, antibodies—making them an essential tool in scientific research and medicine.
Thanks to their specificity and versatility, monoclonal antibodies generated through hybridoma technology play a key role in various applications, from pre-clinical research and diagnostic assays to flow cytometry, immunohistochemistry (IHC), and even cutting-edge therapeutic treatments.
Key Benefits to Monoclonal Antibodies Developed through Hybridoma Technology
- Consistent & Predictable Results
- Exceptional Sensitivity and Specificity
- Versatility in In vitro and In vivo Applications
- Steady and Reliable Supply Chain
- Success with Challenging Targets
Explore More Services
- Mouse Monoclonal Antibodies: Hybridoma Development
- Polyclonal Antibody Production
- Anti-Idiotypic Antibodies
- Repetitive Immunizations Multiple Sites (RIMMS)

Why Choose Green Mountain Antibodies?
Experience You Can Trust – With nearly 30 years of expertise in developing mouse monoclonal antibodies, we’ve worked with all types of antigens and complex screening parameters. Our optimized protocols are designed to quickly produce highly specific, high-affinity antibodies—helping you get results faster.
Save Time, Accelerate Results – Our streamlined protocol and collaborative data review process ensure smooth progress at every stage. By discussing fusion and subcloning screening data the same day, we eliminate unnecessary delays—saving you weeks in your hybridoma development process.
Personalized Support – You’ll have a dedicated project manager guiding you every step of the way, ensuring clear communication, tailored project design, and seamless coordination to help you achieve your goals.
Reliable US-Based Manufacturing – Partnering with a US-based company means dependable quality, safety, and efficiency. Plus, our budget-conscious locked-in pricing helps you avoid unexpected costs and delays.
Flexibility & Innovation – We love exploring new ideas and approaches! Whether you have a unique request or want to brainstorm innovative solutions, we’re always eager to collaborate and learn from our customers to better align with project goals and customers’ unique workflows.
At GMAb, we’re more than just a service provider—we’re your research partner. Let’s create something great together!
Let’s Bring Your Project to Life
At Green Mountain Antibodies, we want to fully understand your project—its goals, target, end application, and timeline. The more details you share, the better we can strategize and deliver the best results for you.
Getting Started – Reach out to us at sales@greenmoab.com to review our Non-disclosure Agreement (NDA), or download the “Start a Project” form and send it our way.
Antigen Design – Whether you have reagents ready to go or need expert input on peptide design or protein selection our experienced science team will help guide the selection of the best immunizing antigen for your project.
Project Planning – We’ll begin with a detailed discussion of your project, followed by a customized Project Plan and budget for your approval.
Hybridoma Development & Assay Services – Once the signed Project Plan, reagents, and PO are in place, your project moves onto our schedule. Our goal? To get started within a week after receiving materials. The immunization, fusion, and subcloning process typically takes 3 to 4 months.
We’re here to make the process smooth, efficient, and tailored to your needs. Let’s work together to create the monoclonal antibodies you need!
1. Phase I: Immunization and Serum Screening
This phase typically spans 3 to 6 weeks. It involves introducing a protein or peptide-based antigen, often requiring 2 –3 mg of soluble antigen, to stimulate an immune response.
2. Phase II: Fusion and Screening of Parental Cultures
Over 17-18 days, splenocytes and/or lymph nodes are fused with myeloma cells to create hybridomas. This stage focuses on the initial screening of parental cultures for viable hybridoma cells that are producing antibodies specific to your reagents.
3. Phase III: Subcloning and Screening
Taking 4 weeks, subcloning refines the selection of hybridomas to ensure cell line stability, specificity, and monoclonality in advance of antibody production.
4. Phase IV: In Vitro Monoclonal Antibody Production
This phase lasts 4-6 weeks and involves the production and characterization of monoclonal antibodies under controlled conditions.
Assay Options to Fit Your Project Needs
We offer a range of assay options to ensure your monoclonal antibodies are optimized for your specific application. If your project could benefit from flow cytometry, Western blotting, or immunohistochemistry (IHC), we can incorporate these into the screening process at each phase.
ELISA – Our Primary Screening Method
ELISA is our go-to screening technique, providing high specificity and sensitivity for detecting and quantifying proteins
Western Blotting
If your end application requires protein identification in a sample, Western blotting may be the better option, offering a reliable way to detect target proteins.
Flow Cytometry
Monoclonal antibodies play a key role in characterizing and sorting cell populations based on specific markers, enabling detailed cellular analysis.
Immunohistochemistry (IHC)
For tissue-specific studies, IHC allows antibodies to visualize the distribution and localization of target proteins within tissue sections—providing valuable biological context.
Not sure which assay is right for your project? Our team is happy to help you choose the best approach to meet your research goals.
Once purified material is available, we can perform additional characterization to further refine your monoclonal antibodies.
Easy, On-Demand Data Access – All project data is securely shared through Citrix ShareFile, giving you instant access whenever you need it.
Continuous Cell Culture – To keep things moving smoothly, cells remain actively cultured throughout the process, eliminating delays and avoiding freeze/thaw cycles.
We’re committed to making your antibody development process as efficient and transparent as possible!
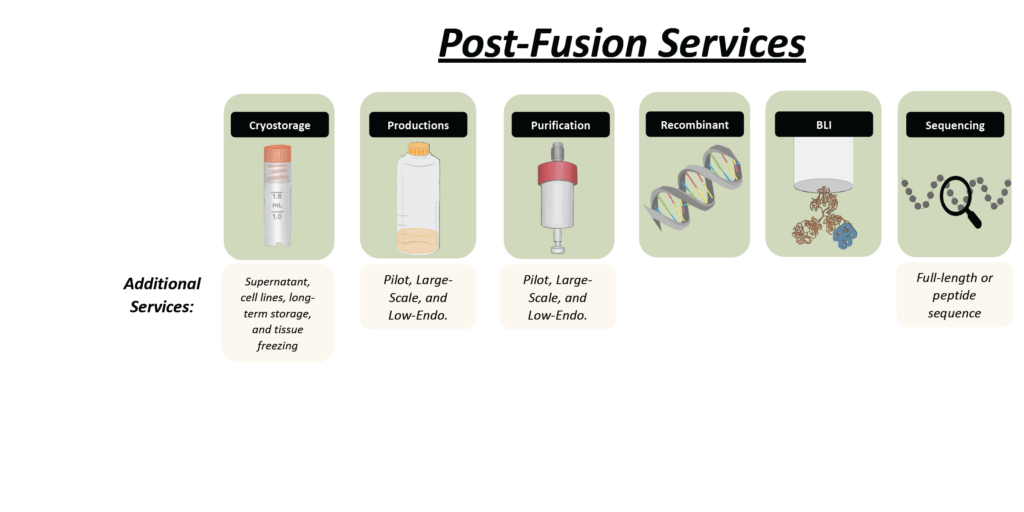
Antibody Production, Purification & Assay Services
Once we successfully develop a panel of stable hybridoma cell lines, we take the next step in ensuring high-quality monoclonal antibodies for your research.
Sequencing & Characterization – Secreted monoclonal antibodies are sequenced and characterized from small-scale productions to ensure reliability and consistency.
Scalable Production – Need more material? We can produce milligram to gram quantities using in vitro methods, with an estimated timeline of 4–6 weeks.
Comprehensive Quality Assurance and Control – Our robust QA and QC testing includes:
✔ Size exclusion chromatography (SEC) – For purity analysis
✔ Dynamic light scattering (DLS) – To assess particle size and stability
✔ Isotype & Isoelectric focusing – For antibody classification
✔ SDS-PAGE – To evaluate protein integrity
✔ Stability testing – Ensuring long-term reliability
✔ Antibody sequencing – Providing full molecular insights
✔ Affinity & Pairing (Gator analysis) – Using biolayer interferometry for precise binding assessment
Our goal is to deliver high-quality, well-characterized antibodies tailored to your needs—efficiently and reliably!
Additional Custom Services
- Transient Antibody Expression: Offers rapid antibody production without the need for long-term cell line development.
- Custom IVD Antibodies and Protein Production Services: Tailored solutions for in vitro diagnostic needs, providing specific antibodies.
- Immunoassay Development Services: Expertise in designing and developing assays for precise protein or antibody screening during hybridoma development.
- Antibody Conjugation Services: Specialized in attaching antibodies to various molecules to enhance their functionality, including biotin, ruthenium and alexafluors.
- Monoclonal Antibody Manufacturing: Focus on producing uniform, high-quality monoclonal antibodies for consistent results.
- Cell Banking, Optimization, and Long-Term Storage: Ensures reliable multi-site monitoring and cell line maintenance for optimal viability and antibody production.
- Protein Purification Services: Delivers pure antibodies or proteins, essential for research and therapeutics.
- Polyclonal Antibody Production: Robust polyclonal antibody production, leveraging expertise to produce antibodies with high specificity and affinity. Performed concurrently with a monoclonal development project.